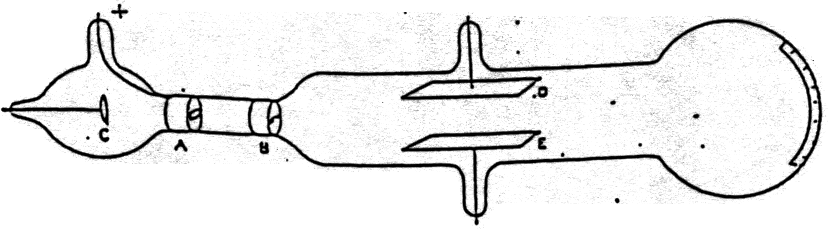
 Thomson also concluded that the cathode rays were made up of negatively charged particles that he called electrons. However, by the late 1890s, he began conducting experiments using a cathode ray tube known as the Crookes’ Tube. Thomson concluded that the cathode rays were negatively charged and were being repelled by the metal plate. When the magnet was placed near the anode, the cathode rays were deflected away from the anode. When the magnet was placed near the cathode, the cathode rays were deflected towards the anode. Thomson used a magnet to deflect the cathode rays. Thomson’s experiment consisted of a glass tube with a cathode and an anode, and a metal plate between the cathode and anode.Ritter concluded that the cathode rays were negatively charged and were being repelled by the metal plate. Ritter observed that when he placed a metal plate between a negatively charged cathode and an anode in a glass tube, the intensity of the light emitted by the tube decreased. Cathode rays were discovered by Johann Wilhelm Ritter in 1802. Thomson’s cathode ray experiment was designed to determine the nature of cathode rays.The electron configuration of a compound ion can be different from the electron configurations of the individual atoms that make up the ion. 
Compound ions are formed when two or more atoms join together to share electrons.When an atom becomes an ion, the number of protons in the nucleus remains the same, but the number of electrons changes.An atom becomes a negative ion (anion) when it gains electrons.An atom becomes a positive ion (cation) when it loses electrons.When an atom gains or loses electrons, it becomes an ion. During the experiment, he discovered the electron, which is one of the. The electron configuration of an atom can be changed by adding or removing electrons. One of the phenomenal contributions of Thomson to science is his cathode ray experiment.The number of electrons in an atom can also be called the electron configuration. The number of protons in a nucleus is also called the atomic number. The number of electrons in an atom is equal to the number of protons in the nucleus.It is one of the most common particles in the universe. The electron is a stable particle that does not change over time.It is a tiny particle, with a diameter of about 2.8 × 10-15 meters. The electron has a mass that is about 1/1837 the mass of a proton. 
It is one of the fundamental particles that make up atoms.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
